3. The AMNOG procedure: more than just cost control
In Germany, there is no fourth hurdle for new drugs. Health economic evaluations does not play an important role in early benefit assessments based on the German Act on the Reform of the Market for Medicinal Products (AMNOG), nor are such evaluations intended to be a regulatory threshold for market access. Therefore, no drug is currently denied access to the market in Germany because of a poor cost-benefit ratio.
The primary goal of the AMNOG procedure is therefore not to limit market access, but to control costs. Until AMNOG came into effect in 2011, pharmaceutical companies were relatively free to set the prices of new drugs - without proof of benefit. Early benefit assessments of new drugs make coverage of costs after the first six months on the market dependent on the proven added benefit versus the so-called appropriate comparator therapy, i.e. standard treatment. If this cannot be proven, only the price that the statutory health insurance (SHI) funds pay for the standard drug can be covered. In its AMNOG report for 2019, one of the large German SHI funds, the DAK, stated that the annual savings for the SHI amounted to more than three billion euros.
Beyond the benefit-based pricing of patent-protected drugs, AMNOG has further positive effects compared to the situation in other countries: Since every new drug in Germany must undergo the AMNOG procedure, an independent body, IQWiG, assesses the patient-relevant benefit of every drug with the exception of orphan drugs, i.e. drugs for the treatment of rare diseases. For orphan drugs, an added benefit is legally deemed to exist, and the decision-making body in the SHI system, the Federal Joint Committee (G-BA) decides on the extent of added benefit.
The AMNOG procedure enables physicians to obtain detailed information on the benefits and harms of the new drug before prescribing it to patients. At the same time, it creates a degree of transparency that hardly exists in other countries. For example, pharmaceutical companies are obliged to disclose their study results in the dossiers on new drugs submitted to the G-BA. The transparency principle also applies to the G-BA and IQWiG. Both IQWiG's dossier assessments and the G-BA's decisions are public, as is the decision-making process in the plenum before the decisions are made.
Even though cost control is a main focus of AMNOG, the procedure also has an influence on the extent to which a drug establishes itself on the market. This is because physicians can decide which drug to prescribe to patients based on freely available and independent information about its mode of action, added benefit and harm.
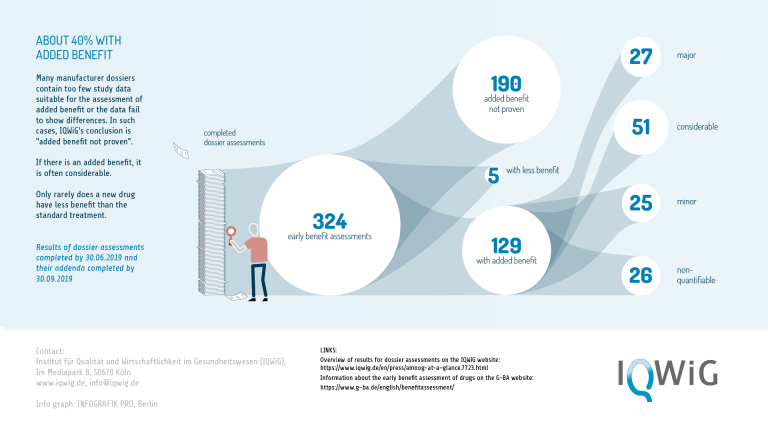
Finally, detailed analyses of early benefit assessments provide starting points that can also be used to improve EMA's approval process. For example, an analysis of 216 IQWiG early benefit assessments of new drugs entering the market between 2011 and 2017 (Wieseler et al. 2019), almost all of which were approved by the EMA for the European market, shows that no added benefit could be determined for more than half of them. Only 54 (25%) of the assessments showed a major or considerable added benefit. Such analyses can initiate a discussion on how the number of genuine innovations in the pharmaceutical sector could be noticeably increased, not only in Germany but also in the whole of the EU.