2. Drug approval and benefit assessment in other countries
Drug approval
In Germany, a pharmaceutical company's option to market a new drug with coverage of costs by the SHI funds initially depends only on the drug's approval.
In other EU countries, but also in further countries such as the USA, the requirements of the approval process are very similar in many areas: A new drug is tested for pharmaceutical quality, safety and efficacy as well as tolerability. In controlled studies, the drug must at least be better than a placebo. Deviations from this scheme exist, for example, in the case of accelerated approval procedures, which can only be pursued under special conditions.
An important step towards the international harmonization of approval procedures beyond the EU was the introduction of a Common Technical Document (CTD) to apply for approval. This has been mandatory in Europe, Japan and the USA since 2003.
Despite this harmonization, it is possible for national authorities to reach different conclusions. For example, a drug may be approved in the USA but not in the EU. This is, among other things, due to that fact that there is room for interpretation as to whether a drug has a good pharmaceutical quality and is safe, effective and tolerable.
Benefit assessment
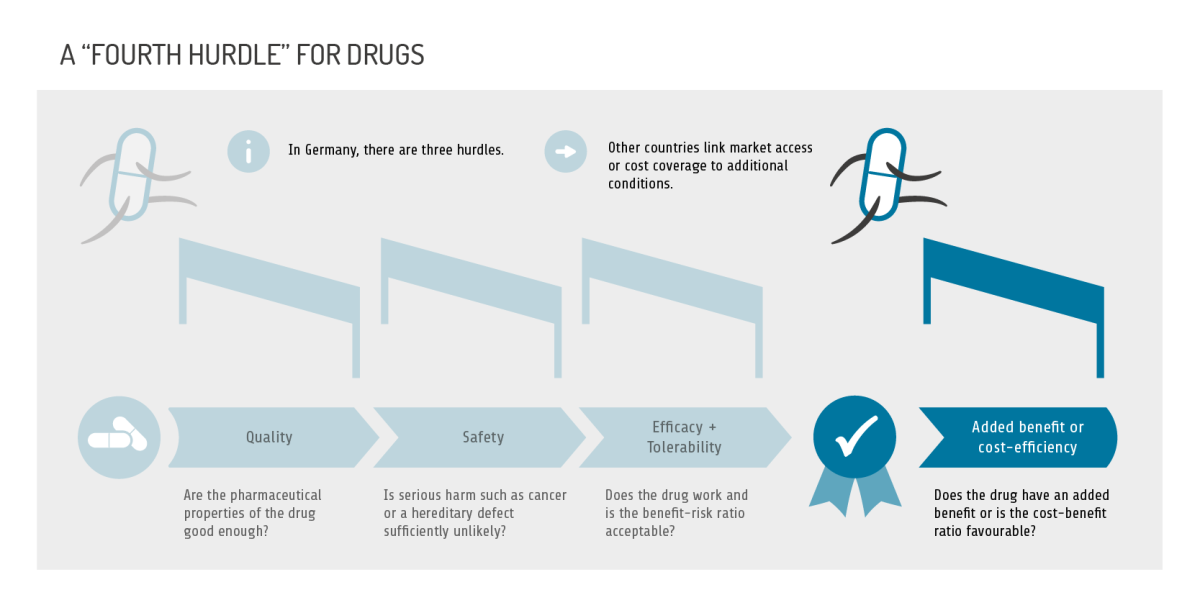
Over the past three decades, countries have developed additional parameters and processes for deciding on market access and/or coverage that go beyond the three hurdles of quality, safety, and efficacy/tolerability.
This is based, on the one hand, on the fact that payers are confronted with ever-increasing health care costs, while resources are limited. On the other, although pharmaceutical companies usually claim that new drugs are better than existing ones and therefore justifiably more expensive, as research has repeatedly shown, these claims are often not confirmed ("bogus innovation"). Most studies even put the proportion of real innovations at less than 15 percent.
Health care systems have responded with two approaches, some of which are intertwined. In one group of countries, the decision on market access and coverage is based on purely medical parameters of benefit. In addition to the conventional approval procedure, new drugs undergo further procedures (such as the early benefit assessment in Germany already described) in which a drug must prove its postulated added benefit versus standard treatment and not just versus placebo. The data from these comparative studies must demonstrate an added benefit in patient-relevant outcomes, such as a decrease in mortality or in the burden of disease (morbidity) or an increase in quality of life. Higher costs claimed by the pharmaceutical company will only be reimbursed if an added benefit is actually proven.
Countries with this approach are, for example, France, Austria, Denmark, Italy, Spain, Switzerland, Turkey, and South Korea.
Cost-effectiveness
In a second group of countries, not only purely medical parameters play a role; coverage decisions are also made on the basis of health economic analyses. The first country to include cost-effectiveness analyses in the decision-making process was Australia in 1993, followed by Canada in 1994. Other countries applying this approach are England, Wales, Scotland and New Zealand.
Some countries use a threshold, i.e., a ratio of costs per additional unit of benefit that may not be exceeded. The quality-adjusted life year ( QALY) is often used as the unit of benefit in the denominator of this ratio. This measure combines qualitative assessments of health conditions with quantitative information on the duration of these conditions. Prolonging survival or improving health-related quality of life increases the value; shortening life or impairing quality of life decreases it. One advantage of the QALY is that the measure is generic, making the benefits of treatments comparable across different therapeutic indications.
However, this procedure is not performed with the same degree of rigor in each country. In Australia, exceeding the threshold can lead to a drug not being included in the positive list, i.e., the list of drugs whose costs are covered by the national health care system. In the Netherlands, even if the cost-benefit ratio is very unfavourable, reimbursement is almost never refused. And in England, after a cost-effectiveness analysis with a negative result, alternative coverage options may be offered, often after a lively public debate.
In Germany, in general no cost-effectiveness analyses are performed for new drugs. However, if SHI bodies and pharmaceutical companies fail to agree on coverage during price negotiations, health economic parameters can also play a role in order to reach an agreement. However, thresholds are not used in this context.
The fourth hurdle
If market access or coverage of a drug depends on additional criteria besides quality, safety and efficacy/tolerability, this is referred to as a "fourth hurdle". In some countries, such as France, this consists of proof of added benefit, while in others, such as Australia, it consists of proof of cost-effectiveness.
The early benefit assessment in Germany is not a fourth hurdle, since drugs without proven added benefit can remain on the market. However, the cost of such drugs should then be no higher than the cost of standard treatment.
Special case (USA)
In the USA, after approval by the Food and Drug Administration (FDA), the list price of a new drug is set by the pharmaceutical company alone; in Germany this is only possible for the first year after approval. There is no government body or other institution in the USA that could negotiate or set this price for the entire country. Instead, numerous private health insurance funds, pharmacy or hospital chains and wholesalers negotiate discounts for new drugs directly with the pharmaceutical companies - sometimes through third-party pharmacy benefit managers. Pharmaceutical companies can also increase the prices of their drugs on an annual or even semi-annual basis. This led, for example, to the price of more than three-quarters of best-selling drugs increasing by more than 50 percent from 2012 to 2016, sometimes for no apparent clinical reason (see Robinson 2020).
Publicly funded health care schemes such as Medicare (for the elderly and disabled), Medicaid (for people with low income), and the US Army Veterans' Health Insurance Program also negotiate discounts with pharmaceutical companies. While costs are not a factor at all in Medicare, the Veterans' Health Insurance requires cost-effectiveness analyses, which may or may not be based on QALYs.
The approach in the USA contributes to the fact that the cost of drugs is higher than in any comparable country, such as other OECD countries. Proponents argue that it is only because of the high drug prices that it is possible to develop so many innovative drugs in the USA.
New drugs: approval, benefit assessment, coverage
1. Drug approval and early benefit assessment in Germany
2. Drug approval and benefit assessment in other countries
3. The AMNOG procedure: more than just cost control