Results
The results of IQWiG's work differ in scope, objective and target group. For example, we develop recommendations to support policy decisions by the Federal Joint Committee (G-BA), as well as easily understandable health information for the general public.
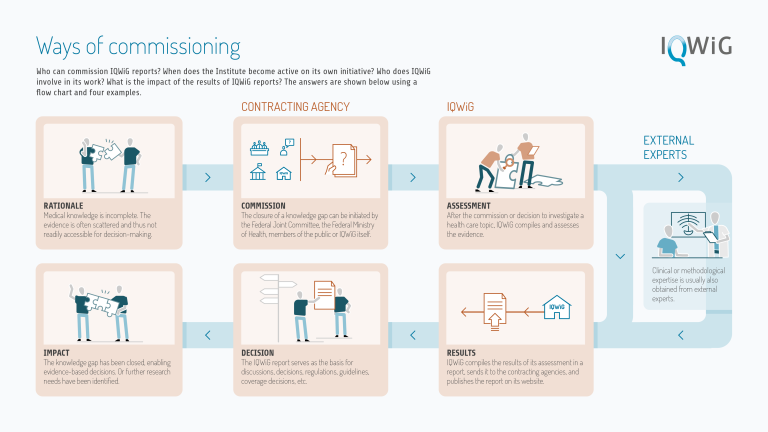
IQWiG usually receives commissions from the G-BA and (less frequently) the Federal Ministry of Health (BMG).
IQWiG produces:
Reports
Detailed reports on benefit assessments, health economic evaluations or guideline synopses. Interim results are also published; interested parties can comment.
Rapid reports
Rapid reports provide timely information on relevant current topics. In this accelerated procedure, no interim products are published, only the final document.
Dossier assessments
Dossier assessments within the framework of the early benefit assessment of drugs (according to AMNOG). Comprehensive dossiers submitted by pharmaceutical companies to the G-BA form the basis of the assessments. IQWiG examines whether the dossier proves an added benefit of a drug versus an appropriate comparator therapy.
Assessments of potential
Assessments of potential in the framework of the testing regulation (according to GKV-VStG). Applications for testing submitted by the manufacturer or provider to the G-BA form the basis of the assessments. The G-BA commissions IQWiG to examine the testing potential of examination or treatment methods that are not solely dependent on the use of drugs (non-drug interventions). This includes medical devices.
Assessments according to §137h
Assessments according to §137h SGB V within the framework of assessments of new treatment and examination methods with high-risk class medical devices (according to GKV-VSG). Documents transferred to the G-BA by hospitals and, if applicable, by medical device manufacturers concerned form the basis of such an assessment. On behalf of the G-BA, IQWiG assesses the benefit and the potential of the method.
ThemenCheck reports
ThemenCheck reports are based on proposals made by members of the public in the “Themencheck Medizin" (Topic check medicine) for assessments of examination and treatment procedures. The ThemenCheck reports always include an assessment of the benefit and harm of a medical intervention. In addition, depending on the respective topic, further HTA-relevant aspects (economic, ethical, legal, social, and organizational) are covered to a varying degree.
Health information
Health information: Easily understandable information for the general public to help make informed decisions on individual health issues. You will find this information on the website www.informedhealthonline.org
Addenda
Addenda on complete reports, rapid reports or dossier assessments, if further need for work arises after completion of an assessment. For instance, this might be the case if the evidence base changes during the consultations of the G-BA, e.g. if new studies are published.
Working papers
Working papers on relevant developments in health care and on the methodological work of the Institute.