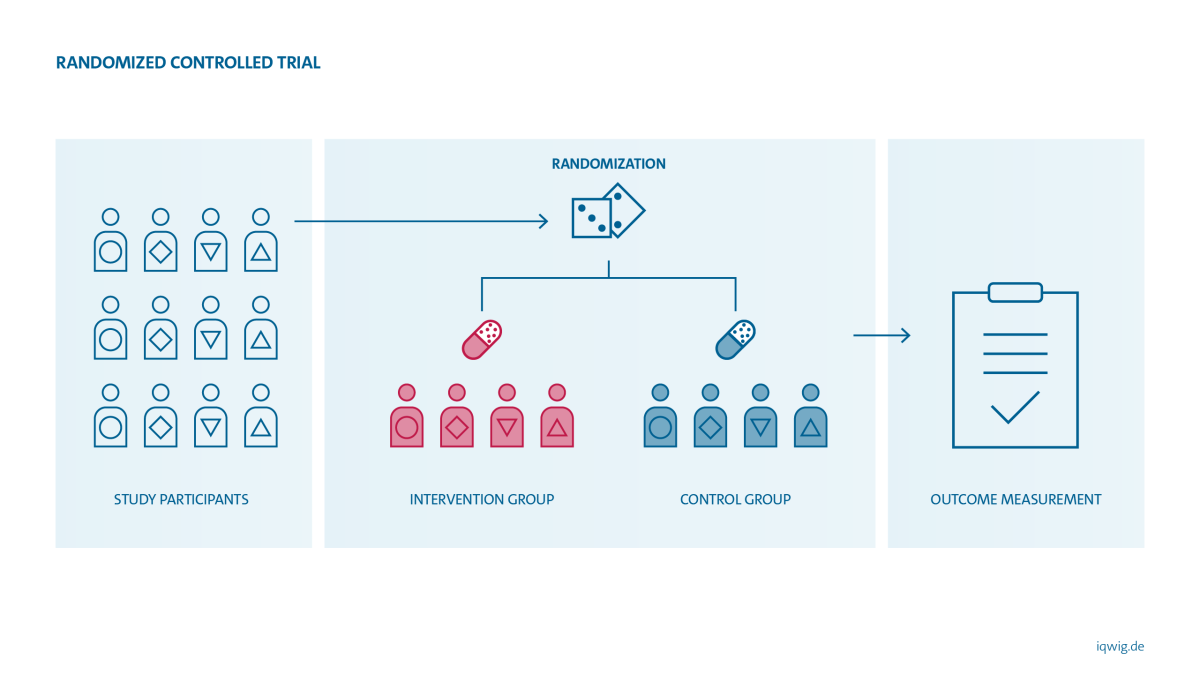
Why are randomized controlled trials (RCTs) considered the gold standard?
The design of a study has a decisive influence on the reliability of its results. Whether the observed effect (or side effect) is actually attributable to the drug used can only be determined if other influencing factors are completely or largely excluded. This is possible if patients are randomly assigned to treatment groups (= randomization). Potentially influencing factors such as age, gender, comorbidities or lifestyle are thus randomly and evenly distributed across the groups. The only difference between the treatment groups in the study should then be the treatment used. If a difference in disease severity, symptoms or side effects is observed between the treatment groups during the course of the study, the difference can be attributed with greater certainty to the effect of the treatment.
In almost all cases, drugs and other treatment methods evaluated by IQWiG can be studied in RCTs. IQWiG therefore generally uses RCTs in its benefit assessments. Only in well-justified exceptional cases does it resort to non-randomized intervention studies or observational studies. Such exceptions may exist if other types of studies also provide sufficient certainty of results for the question at hand. In the case of diseases that are rapidly fatal without treatment, for example, several consistent case studies may show that a particular intervention prevents this course of events – and provide sufficient certainty of results. In some cases, it is possible to reliably evaluate diagnostic tests even in the absence of RCTs.
Fundamental aspects of benefit assessments explained simply
IQWiG's methods are described in detail in its methods paper. But why does IQWiG work the way it does? What is so important about a direct comparison? And what exactly is the appropriate comparator therapy? We explain these and other aspects of our methods briefly on these pages.