Why do we need comparative data for benefit assessments?
In medicine, decisions often involve choosing between two alternatives: these may be treatments, complex lifestyle changes, watchful waiting, or diagnostic procedures and screening tests. The question always arises: is Option A better than Option B?
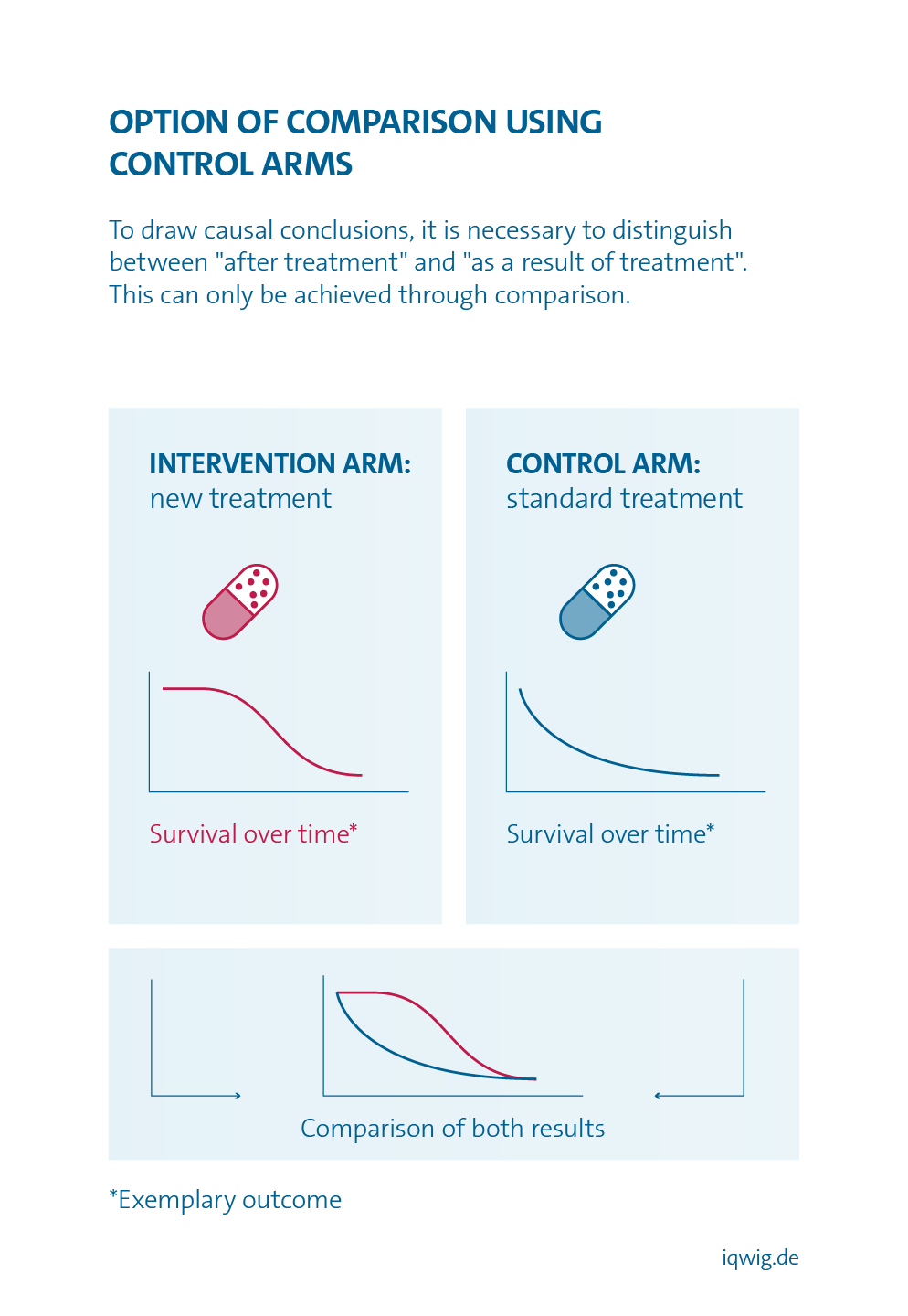
If there are only results for one treatment option, it is only possible to determine whether patients felt better after treatment than before. However, it is not possible to conclude with certainty from such an observation whether the improvement was actually due to the treatment. Even with an ineffective treatment option, patients may feel better after treatment than before, e.g. due to a placebo effect. It is therefore necessary to compare two groups, one of which received one option and the other the other option. The most reliable method is to compare treatments in direct comparative, randomized studies (randomized controlled trials).
Fundamental aspects of benefit assessments explained simply
IQWiG's methods are described in detail in its methods paper. But why does IQWiG work the way it does? What is so important about a direct comparison? And what exactly is the appropriate comparator therapy? We explain these and other aspects of our methods briefly on these pages.