What is the appropriate comparator therapy (“zweckmäßige Vergleichstherapie“, zVT)?
The appropriate comparator therapy is the treatment with which a new drug is compared in the early benefit assessment in accordance with the German Act on the Reform of the Market for Medicinal Products (AMNOG). The question addressed in the early benefit assessment is whether the drug under investigation is better than the appropriate comparator therapy and thus offers an added benefit.
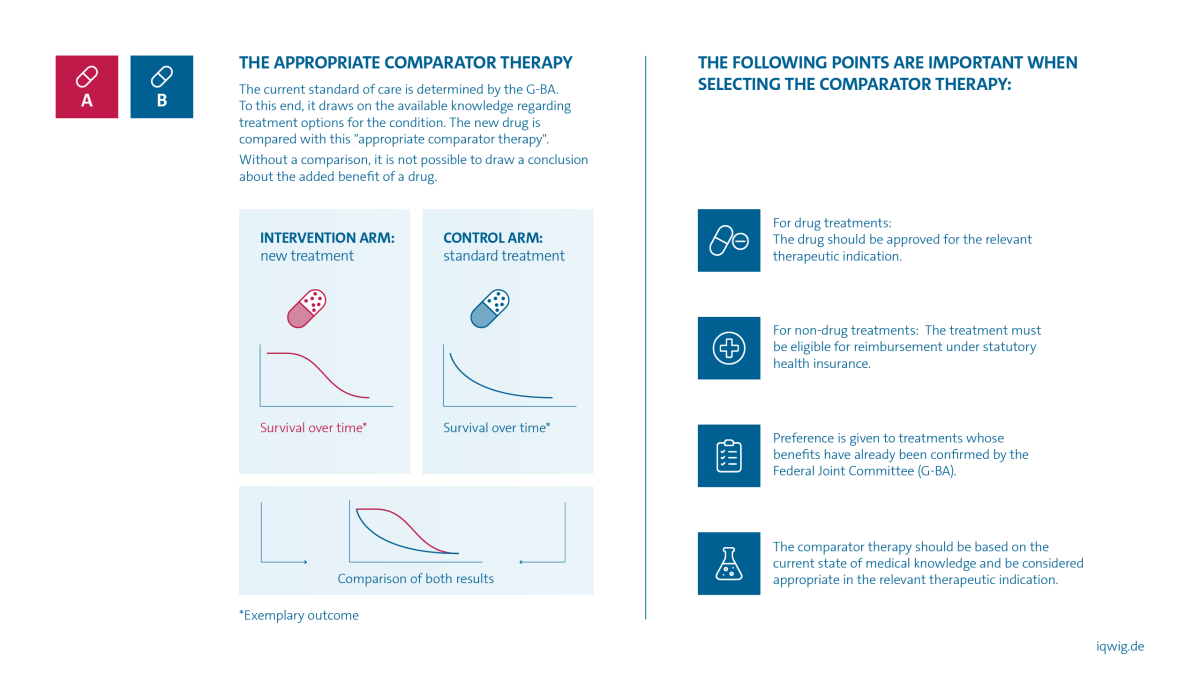
The appropriate comparator therapy is determined by the Federal Joint Committee (G-BA). This involves one or more therapies that are common and appropriate in the respective medical field. They represent the current standard of care.
When determining this comparator therapy, the G-BA is guided by international standards of evidence-based medicine, i.e. scientifically proven findings. Ideally, there are studies that prove the effectiveness of this treatment, and it has proven itself in practice. If clinical guidelines exist, these can influence the choice of the comparator therapy.
The appropriate comparator therapy does not necessarily have to be a drug. It can also consist of supportive measures or pure observation without active treatment.
The following points are important when selecting the comparator therapy:
- For drug treatments: The drug should be approved for the respective disease.
- For non-drug treatments: It must be possible to reimburse the treatment through statutory health insurance.
- Preference is given to treatments whose benefits have already been confirmed by the G-BA.
- The comparator therapy should be based on the current state of medical knowledge and be considered appropriate for the respective disease.
Fundamental aspects of benefit assessments explained simply
IQWiG's methods are described in detail in its methods paper. But why does IQWiG work the way it does? What is so important about a direct comparison? And what exactly is the appropriate comparator therapy? We explain these and other aspects of our methods briefly on these pages.